|
3/16/2023 0 Comments Ph when you mix base with water  
And, none of the results are toxic, hence, we can do it at home safely. Herefore, when vinegar and baking soda is mixed, the results are sodium acetate, carbon dioxide, and water. This particular reaction always forms water and carbon dioxide as by-products. When they react, they form a new compound through the neutralization reaction. Vinegar (CH 3COOH) and Baking Soda (NaHCO 3) are both weak acids and weak bases, respectively. The resulting mixture can be used for cleaning, beauty purposes, etc. Since both of them are weak acids and bases, we can mix them without worrying about the consequences, as it’s not causing any harm to ourselves or our property when we do it at home. However, let us learn if this method really works as we think it does and learn more interesting facts about the vinegar and baking soda solution. One of those examples is the use of vinegar and baking soda mixture for many household chores, starting from cleaning hard water stains to clearing clogged drains, and what-not. Just like we’re moving from eating processed food to organic ones, we’re also replacing our use of chemical elements in our households with organic elements. This is because, every chemical-based element has a way of harming us, and our society and world as a whole–if not immediately, then after a long time. It may include cleaning, consuming, wearing clothes, using bags, etc. Without this buffer system, the pH in our bodies would fluctuate too much and we would fail to survive.As more time goes by, people are moving from toxic, chemical-based materials to organic-based ones, irrespective of the purpose. While carbonic acid is an important product in this reaction, its presence is fleeting because the carbonic acid is released from the body as carbon dioxide gas each time we breathe. The H + ions can combine with the OH – ions, limiting the increase in pH. Likewise, if too much OH – is introduced into the system, carbonic acid will rapidly dissociate into bicarbonate and H + ions. If too much H + enters the body, bicarbonate will combine with the H + to create carbonic acid and limit the decrease in pH. This buffer system involves carbonic acid (H 2CO 3) and bicarbonate (HCO 3 –) anion. Carbon dioxide is part of a prominent buffer system in the human body it keeps the pH within the proper range. Buffers readily absorb excess H + or OH –, keeping the pH of the body carefully maintained in the aforementioned narrow range. So how is it that we can ingest or inhale acidic or basic substances and not die? Buffers are the key. Deviation outside of the pH range can induce coma or even cause death. Cells no longer function properly, and proteins will break down. If the pH of the body is outside of this range, the respiratory system malfunctions, as do other organs in the body. Most cells in our bodies operate within a very narrow window of the pH scale, typically ranging only from 7.2 to 7.6. Sodium hydroxide and many household cleaners are very alkaline and give up OH – rapidly when placed in water, thereby raising the pH. The OH – ions combine with H + to produce water, which raises a substance’s pH. Conversely, bases are those substances that readily donate OH –. For example, hydrochloric acid and lemon juice are very acidic and readily give up H + when added to water. The stronger the acid, the more readily it donates H +. Orange juice is mildly acidic (pH = approximately 3.5), whereas baking soda is basic (pH = 9.0).Īcids are substances that provide hydrogen ions (H +) and lower pH, whereas bases provide hydroxide ions (OH –) and raise pH. The environment in your stomach is highly acidic (pH = 1 to 2). The blood in your veins is slightly alkaline (pH = 7.4). Anything below 7.0 (ranging from 0.0 to 6.9) is acidic, and anything above 7.0 (from 7.1 to 14.0) is alkaline. It is neither acidic nor basic, and has a pH of 7.0. Thus, small changes in pH represent large changes in the concentrations of hydrogen ions. A change of one unit on the pH scale represents a change in the concentration of hydrogen ions by a factor of 10, a change in two units represents a change in the concentration of hydrogen ions by a factor of 100. Therefore, the more hydrogen ions present, the lower the pH conversely, the fewer hydrogen ions, the higher the pH. The overall concentration of hydrogen ions is inversely related to its pH and can be measured on the pH scale (Figure 1). High concentrations of hydrogen ions yield a low pH (acidic substances), whereas low levels of hydrogen ions result in a high pH (basic substances). This pH test measures the amount of hydrogen ions that exists in a given solution. 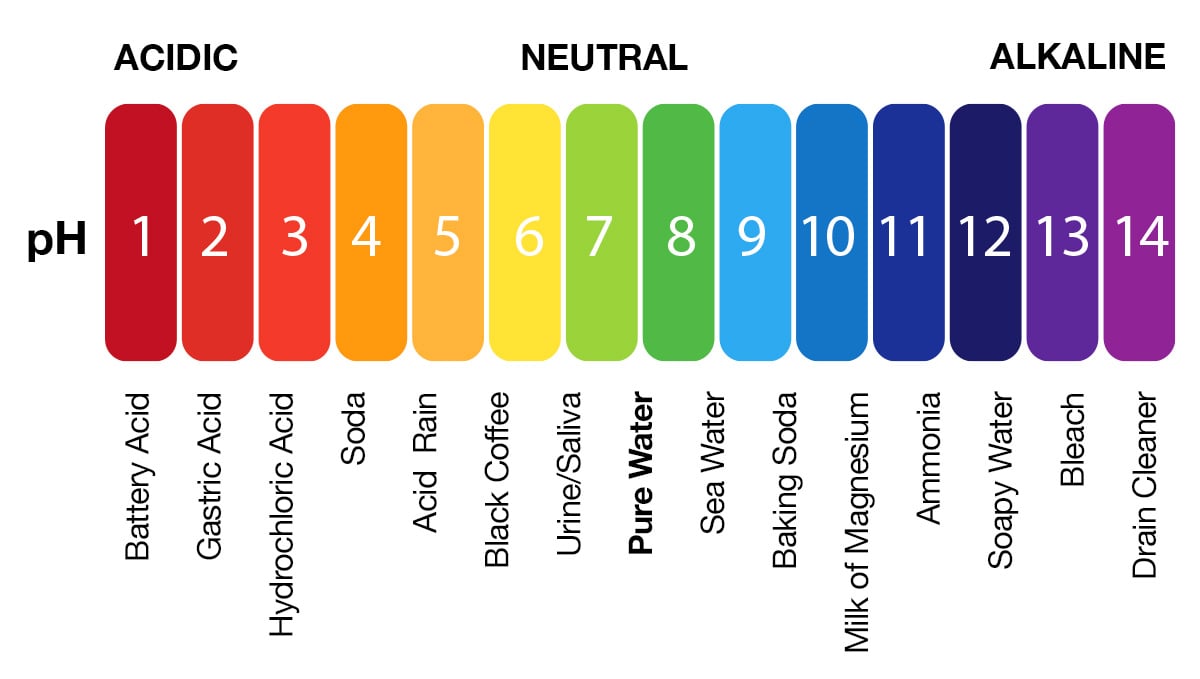
(credit: modification of work by Edward Stevens) 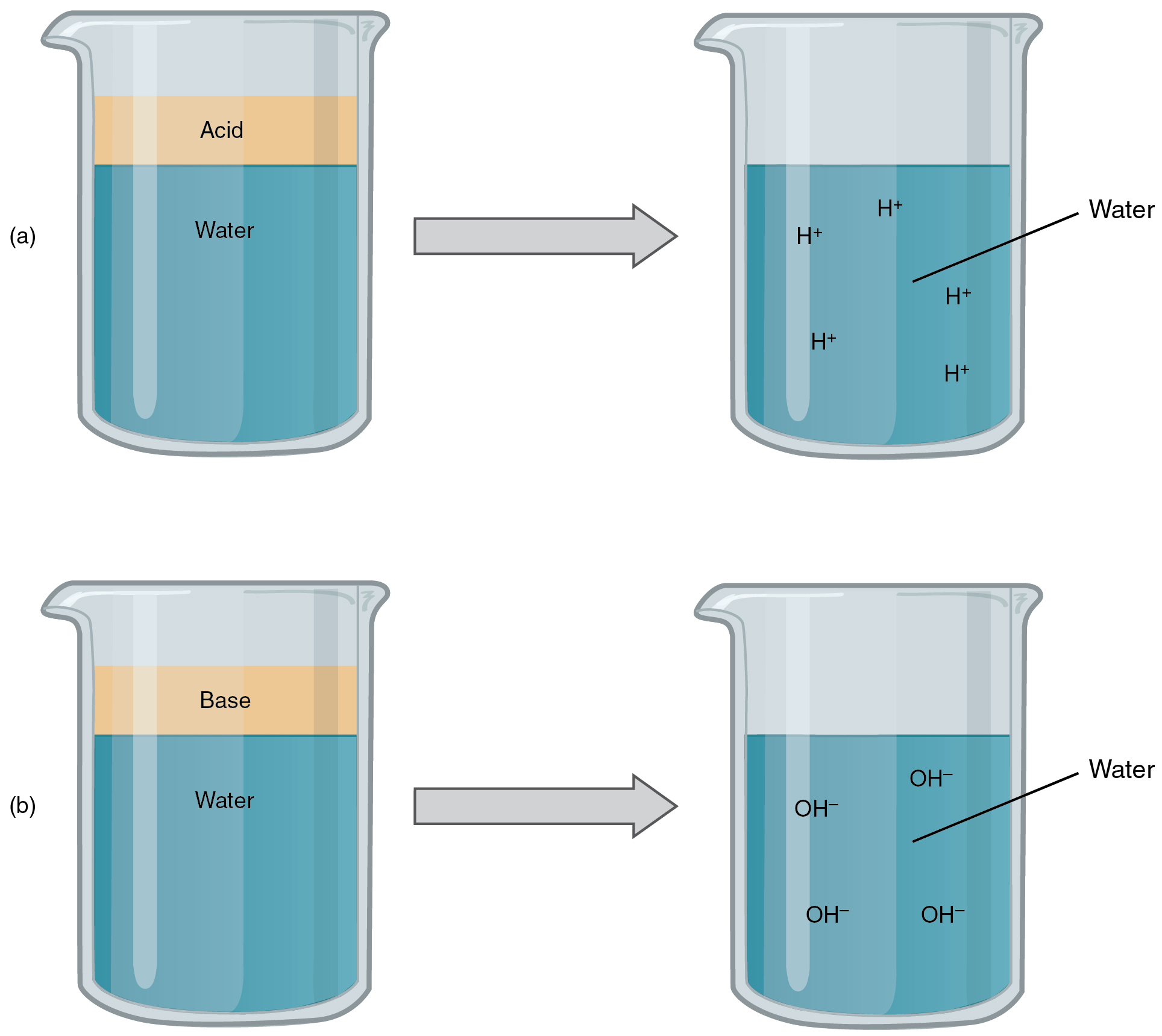
The pH scale measures the amount of hydrogen ions (H +) in a substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
